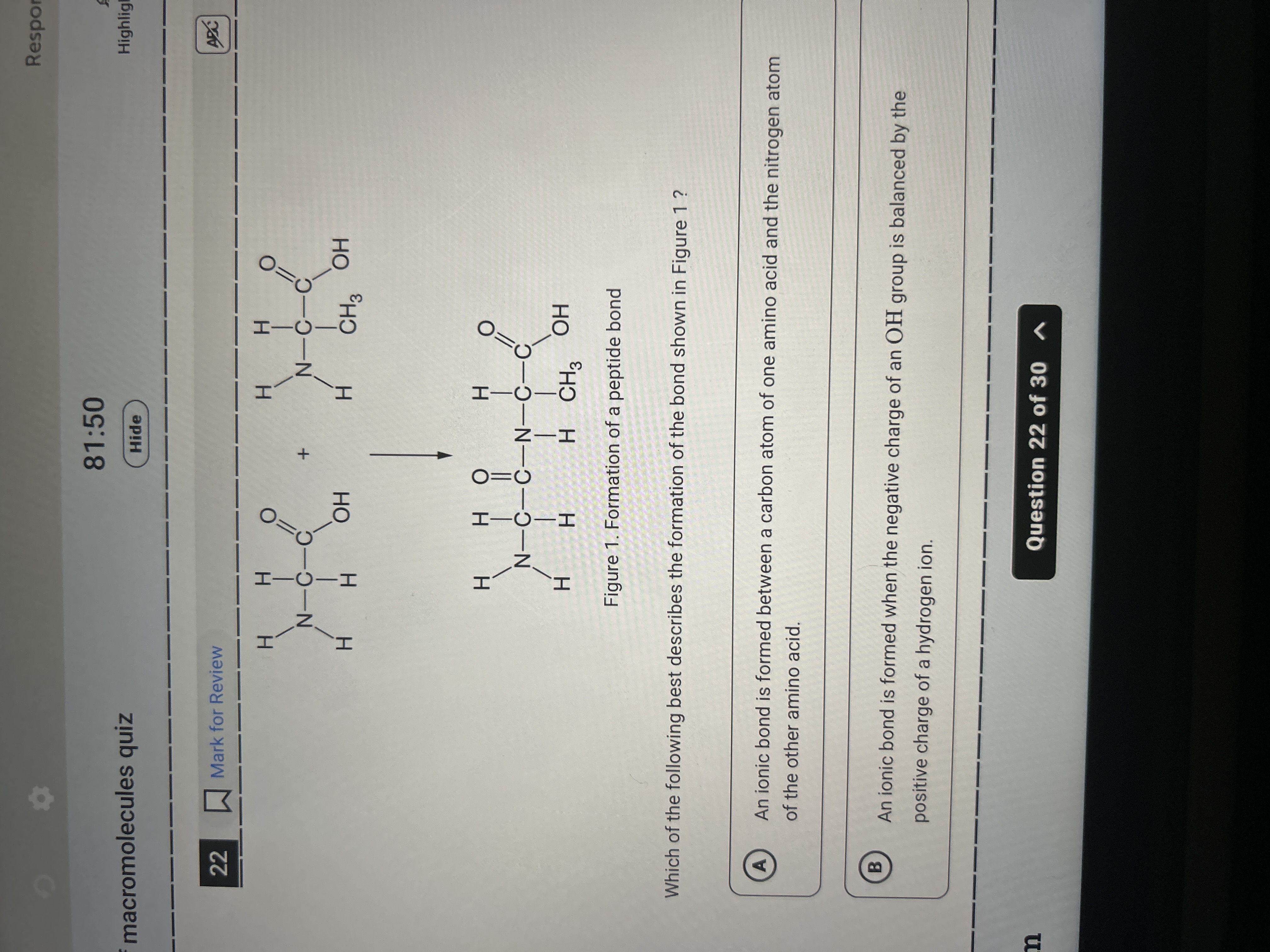
Which of the following best describes the formation of the bond shown in Figure 1?
Understand the Problem
The question is asking for the correct description of the formation of a chemical bond illustrated in the figure. It focuses on discerning the details about ionic bonding in the context of amino acids and peptide bonds.
Answer
Peptide bond formed by dehydration synthesis.
The peptide bond is formed through a dehydration synthesis reaction between the carboxyl group of one amino acid and the amino group of another, resulting in a covalent bond.
Answer for screen readers
The peptide bond is formed through a dehydration synthesis reaction between the carboxyl group of one amino acid and the amino group of another, resulting in a covalent bond.
More Information
The bond shown is a peptide bond, commonly found in proteins, linking amino acids together. This process involves removing a water molecule (H₂O) to form the bond.
Tips
Mistaking an ionic bond for a peptide bond can occur; ensure to recognize the covalent nature and dehydration process of peptide bonds.
Sources
AI-generated content may contain errors. Please verify critical information