Podcast
Questions and Answers
What is the functional group of a carboxylic acid?
What is the functional group of a carboxylic acid?
- OH
- O
- -COOH (correct)
- CO2H
How are aliphatic carboxylic acids named according to IUPAC rules?
How are aliphatic carboxylic acids named according to IUPAC rules?
- Add the prefix hydroxy-
- Drop the final -e and add the suffix -oic acid (correct)
- Add the suffix -oate
- Add the suffix -ic acid
How are other groups bonded to the parent chain named in carboxylic acids?
How are other groups bonded to the parent chain named in carboxylic acids?
- Named using numerical prefixes (correct)
- Named using alphabetical order
- Named using Greek letters
- Named using Latin prefixes
What type of molecule is an aliphatic carboxylic acid?
What type of molecule is an aliphatic carboxylic acid?
What is the solubility of carbons 1-4 in water?
What is the solubility of carbons 1-4 in water?
What type of bonds contribute to the comparatively high boiling points of carboxylic acids?
What type of bonds contribute to the comparatively high boiling points of carboxylic acids?
What type of acids are carboxylic acids generally considered to be?
What type of acids are carboxylic acids generally considered to be?
What is the classification of carboxylic acids that contain one or more C=C bonds?
What is the classification of carboxylic acids that contain one or more C=C bonds?
In which type of carboxylic acid is the carbon of the carboxyl group bonded directly to a carbon in an aromatic ring?
In which type of carboxylic acid is the carbon of the carboxyl group bonded directly to a carbon in an aromatic ring?
What is the result of the oxidation of alkyl groups attached to aromatic rings?
What is the result of the oxidation of alkyl groups attached to aromatic rings?
What type of reactions do acid chlorides, acid anhydrides, esters, and amides participate in?
What type of reactions do acid chlorides, acid anhydrides, esters, and amides participate in?
What is formed by the reaction of an acid with an alcohol or a phenol?
What is formed by the reaction of an acid with an alcohol or a phenol?
What is the product of the reaction between thionyl chloride and carboxylic acids?
What is the product of the reaction between thionyl chloride and carboxylic acids?
What property of acids causes them to change blue litmus to red?
What property of acids causes them to change blue litmus to red?
What is the solubility of carbons 5-8 in water?
What is the solubility of carbons 5-8 in water?
What is the result of the elimination of water from two molecules of carboxylic acid?
What is the result of the elimination of water from two molecules of carboxylic acid?
Flashcards
Carboxylic acid functional group?
Carboxylic acid functional group?
A -COOH group.
IUPAC naming of aliphatic carboxylic acids?
IUPAC naming of aliphatic carboxylic acids?
Drop the final -e and add the suffix -oic acid.
Naming other groups bonded to carboxylic acid parent chain?
Naming other groups bonded to carboxylic acid parent chain?
They are named using numerical prefixes.
Aliphatic carboxylic acids?
Aliphatic carboxylic acids?
Signup and view all the flashcards
Solubility of C1-C4 carboxylic acids?
Solubility of C1-C4 carboxylic acids?
Signup and view all the flashcards
Bonds contributing to high boiling points of carboxylic acids?
Bonds contributing to high boiling points of carboxylic acids?
Signup and view all the flashcards
Carboxylic acids?
Carboxylic acids?
Signup and view all the flashcards
Carboxylic acids with C=C bonds?
Carboxylic acids with C=C bonds?
Signup and view all the flashcards
Carboxyl group bonded directly to a carbon in an aromatic ring?
Carboxyl group bonded directly to a carbon in an aromatic ring?
Signup and view all the flashcards
Oxidation of alkyl groups on aromatic rings?
Oxidation of alkyl groups on aromatic rings?
Signup and view all the flashcards
Acid chlorides, acid anhydrides, esters, and amides?
Acid chlorides, acid anhydrides, esters, and amides?
Signup and view all the flashcards
Acid + Alcohol/Phenol?
Acid + Alcohol/Phenol?
Signup and view all the flashcards
Product of thionyl chloride + carboxylic acids?
Product of thionyl chloride + carboxylic acids?
Signup and view all the flashcards
Acids cause blue litmus to turn red?
Acids cause blue litmus to turn red?
Signup and view all the flashcards
Solubility of C5-C8 carboxylic acids?
Solubility of C5-C8 carboxylic acids?
Signup and view all the flashcards
Elimination of water from two molecules of carboxylic acid?
Elimination of water from two molecules of carboxylic acid?
Signup and view all the flashcards
Study Notes
Carboxylic Acids
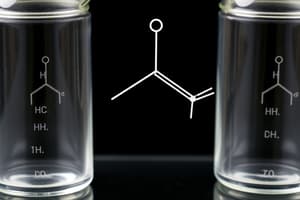
- The functional group of a carboxylic acid is -COOH.
Nomenclature of Aliphatic Carboxylic Acids
- Aliphatic carboxylic acids are named according to IUPAC rules.
- The parent chain is the longest chain that includes the carboxyl (-COOH) group.
Naming Other Groups in Carboxylic Acids
- Other groups bonded to the parent chain are named as substituents.
Characteristics of Aliphatic Carboxylic Acids
- Aliphatic carboxylic acids are organic compounds with a carboxyl (-COOH) group.
- They are polar molecules.
Solubility of Carboxylic Acids
- Carboxylic acids with 1-4 carbons are soluble in water.
- Carboxylic acids with 5-8 carbons are less soluble in water.
Physical Properties of Carboxylic Acids
- The boiling points of carboxylic acids are comparatively high due to hydrogen bonding.
Classification of Carboxylic Acids
- Carboxylic acids are generally considered to be weak acids.
- Carboxylic acids containing one or more C=C bonds are classified as unsaturated.
- Carboxylic acids where the carbon of the carboxyl group is bonded directly to a carbon in an aromatic ring are classified as aromatic.
Reactions of Carboxylic Acids
- The oxidation of alkyl groups attached to aromatic rings results in the formation of carboxylic acids.
- Acid chlorides, acid anhydrides, esters, and amides participate in nucleophilic acyl substitution reactions.
- The reaction of an acid with an alcohol or a phenol forms an ester.
- The reaction between thionyl chloride and carboxylic acids forms an acid chloride.
Acidity of Carboxylic Acids
- Carboxylic acids turn blue litmus to red due to their acidic property.
Condensation Reactions
- The elimination of water from two molecules of carboxylic acid forms an acid anhydride.
Studying That Suits You
Use AI to generate personalized quizzes and flashcards to suit your learning preferences.